Our Work
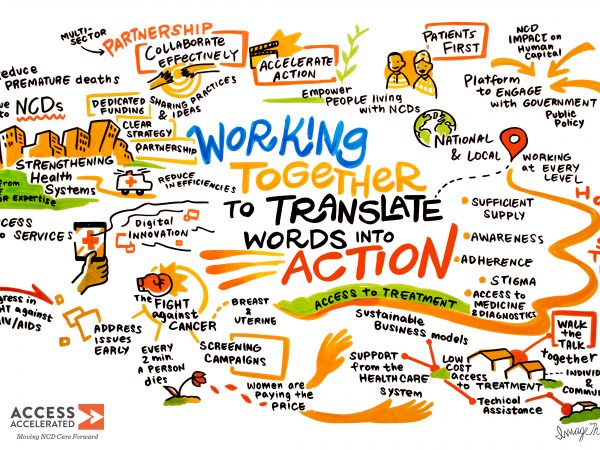
People living with or at risk of NCDs face challenges at every step of their health journey. Our strategy is grounded in the understanding that in order to meet their needs, we must take a people-centered approach. We are currently exploring three areas where we know the holistic perspective brought by uniting the expertise of our member companies can accelerate solutions: supply chain, primary care and digital health.

Access Accelerated exists to transform how the biopharmaceutical industry accelerates global progress on NCDs. Our work to catalyse change, align resources and scale solutions is at the very heart of what we do and informs our three strategic goals.

To achieve these goals we have organized our work into the following action areas.

Guided by our principles, we’re working to support locally defined priorities and develop tailored solutions to ensure scalable and sustainable NCD solutions. We know that for our projects to be successful, they must have strong local ownership and engagement. Our commitment has been to collaborate and communicate with local stakeholders at all stages of program development, execution, and evaluation. This allows us to co-create programs that augment and align with existing systems and build local capacity.
Broadly speaking, our work focuses on three key areas where we know the private sector can help foster sustainable solutions: supply chain, digital health and primary care.
SUPPLY CHAIN
STRENGTHENING SUPPLY CHAINS
Reliable supply chains are vital for health systems to meet population needs. We’re working with partners to close bottlenecks and foster greater efficiencies in low- and middle-income countries.
PRIMARY CARE
Screening for NCDs
Too often, NCDs are diagnosed and treated when symptoms appear or after patients have sought specialty care. Health systems urgently need resources, trained health workers and policies that support early screening.
DIGITAL HEALTH
Leveraging the Power of Technology
Digital health solutions can improve health care delivery and outcomes. Access Accelerated embraces digital service models for NCDs and is working to increase access to quality health care in under-resourced settings using these tools.

Years ago, the future was bleak but now there’s a light at the end of the tunnel. I’m happy that the future is starting to build around partnerships because that’s the only way that we will be able to do something about NCDs.
Dr. Joseph Kibachio, Ministry of Health, Kenya
Terms of Use
IFPMA (“we”, “us”) owns and operates www.accessaccelerated.org (“the/our website”). Any person accessing and/or using the website (“the visitor/user”) agrees to the “Terms of Use”, containing the conditions, rules and other provisions with respect to the access and/or use of our website. If you have questions about the “Terms of Use”, please feel free to contact us at admin@ifpma.org.
CONTENT
General Use
The material contained on this website is for information only. The website is entitled to be served as an information resource for the internal and external stakeholders of the Access Accelerated initiative, including the general public. Third-Party Websites Links to other websites are provided solely as a convenience and therefore imply neither IFPMA’s responsibility for, nor its approval of the information contained in these websites.
Intellectual Property
Any use of information from the website should have an acknowledgement of Access Accelerated/Third-Party as a source respectively, citing the corresponding uniform resource locator (URL).
Logo and Name
The use of Access Accelerated's name and/or logo is prohibited without prior express authorization from IFPMA. The access and/or use of the website does not permit to copy, reproduce, modify, distribute or exploit Access Accelerated's name and/or logo except with the prior explicit permission.
Abuse and Violation
Any abuse or violations of the Terms of Use and/or Privacy Policy should be reported to secretariat@accessaccelerated.org.
DISCLAIMER
IFPMA renounces any liability or responsibility for any damages of any nature whatsoever resulting from the use of, inability to use, and/or reliance on the material and information contained on the Access Accelerated website. IFPMA makes no representations or warranties as to the completeness or accuracy of any of the information available on the website as well as does not provide any warranties regarding the security of the content of our website or any linked websites. IFPMA endeavours to make its best effort to keep the information accurate and updated on a regular basis, however this is not claimed to be exhaustive. IFPMA does not guarantee that the website is continuously available and error-free at all times.
Privacy Policy
IFPMA is committed to protecting the privacy and security of visitors to the Access Accelerated website. Outlined below is our online privacy policy for information collected through our website. If you have questions about these policies, please feel free to contact us at secretariat@ifpma.org.
Personal Information
We constantly analyse our website logs to improve the value of the Access Accelerated website. We do not use this analysis to record or track any personally identifiable information of visitors. If you are asked to provide personal information such as your name, e-mail or/and postal address when requesting information, providing feedback, registering to a newsletter, or using any other functions of the website, be assured that all such information is considered by IFPMA both private and secure. IFPMA does not share or sell your personal information to third parties. Under no circumstance will details be disclosed publicly, except in order to comply with any legal obligation. This policy reiterates that IFPMA fully appreciates the importance of online privacy and is committed to protecting your personal information.
Use of Cookies Files
The Access Accelerated website is enhanced by cookie files to personalize and customize the experience of our visitors and to support some necessary functions. IFPMA collects broad usage statistics using Google Analytics, to better understand how our visitors use our website, and to help us improve visitors’ experience. Such information includes the IP address (where available), browser type, and any additional information provided by a browser during the use of our website. The statistical data about our users’ browsing actions and patterns does not identify any individual. You may choose to accept or refuse cookies by modifying your browser preferences. If you wish to get notified when a cookies are set, this option should also be available in your browser settings. Please note that if you reject some or all cookies, your experience at this and other websites throughout the Internet may not be complete.
Payment Transactions
To enable IFPMA to process any order or registration to an event, a secure online payment system may be used to process payments using credit card details. IFPMA will not be collecting or storing credit card details in our system.
Third-Party Websites
Please be aware that other websites to which links may be provided, including social media, may collect visitor’s personally identifiable information when accessed. The information collection practices of other websites linked to IFPMA’s website are not covered by this Privacy Policy.
Cookie Policy
Some of our web pages utilise “cookies”. A “cookie” is a small text file that may be used, for example, to collect information about web site activity. Some cookies and other technologies may serve to recall Personal Information previously indicated by a web user. Most browsers allow you to control cookies, including whether or not to accept them and how to remove them.
Some cookies are necessary for the operation of our website, if you choose to block them some aspects of the site may not work for you. Non necessary cookies are only set when you have given your explicit consent to their use. Such cookies included those set by our statistics package Google Analytics.
We are committed to privacy and support current industry initiatives to preserve individual privacy rights on the Internet. Protecting your privacy on-line is an evolving area and this website will constantly evolve to meet these demands.